ff
|
How do you feel overall about this class? What characterizes your experience?
Throughout this year Chemistry has changed for me. I didn't know what to expect coming into Chemistry and it was a bit terrifying. I thought it was going to be harder than Spanish, and Spanish was hard for me. But I surprised myself. Chemistry surprised me. I learned so much this semester. I opened myself to lots of new learning experiences and i'm proud for that. The class environment was great for the most part , and I felt welcome. I felt like what-ever I wanted to learn or experiment on, was welcomed. What did you learn about yourself as a learner? Chemistry class has taught me that I work great independently, but also in groups. For most of my projects I grouped up with people. I never liked group work because I would always be the one to do most and sometimes all the work. When I got to be in groups in chemistry, right off the back any group I was in, everyone worked an equal amount. I learned that, for me, when I do work in groups I have to find the right people. Not just my friends all the time. When was a time or what was something you did when you were driven by curiosity? A project I remember doing this semester was the dry ice lab we did way back in the beginning of the semester. I really liked that project and it was the second project we did and I just remember being so fond of how the dry I made the bubbles and when we put food coloring in it and it changed colors.. It was just great. I wanted to try everything and anything with the dry ice. I was curious to where dry ice was made and how it inflated our balloon. Like.. How? One other project was when I made bath bombs with Rebekah. That was by far my one of my favorite projects. I've been wanted to bath bombs forever and to be able to actually make them and them actually working was great. When was a time or what was something that you did when you weren’t driven by curiosity? With the rocket project I liked it but at times I felt like it was a drag on.. I enjoyed it but sometimes I found myself not doing anything because I finished early. After I wanted to start to work on something new, so I searched for different projects I could work on such as making slime. I still didn't find interest in that either. I wish there was another alternative instead of just sitting in class doing nothing.
0 Comments
This weekend I got all scienced up and did some really cool experiments!
For the first experiment, I made rain in a jar. Basically to show how rain happens. It looked really cool and I wanted to try it. So I got moose, food coloring, water, and a glass. I put water in a mason jar and moose on top of the water. I dropped food coloring on the top of the moose and after i little while the food coloring sunk in and it looked like it was raining! Really cool if you ask me. The experiment was mean to show how rain is formed. At the end it kind looked like a lava lamp. For the second experiment, I tried the zip log bag and coloring pencil experiment. So I filled the zip log with water and basically just potted wholes into the bag. I searched up why was it that the zip log didn't break or leak and I found out when a sharp pencil tip pushes between the polymers (a substance in the zip log bag) they stretch tight around its surface. This plastic hug keeps the water sealed inside, and the pressure of the water helps keep the seal in place. So I wasn't damaging the individual molecules the bag is made up of, I was just separating them! Looking Back: What happened? After agreeing on how we wanted our rocket to turn out, we began to make our rocket body. We wrapped the paper, and realized that it might be better to increase the body size from ten inches to about twelve and inches. Then, we made our fins. We estimated the area of the fins, and once we felt like our center of thrust was right around where 50% of the surface area was, we finalized our fin design, and glued the fins onto the rocket. After that we painted our rocket to an ombre look with pink and purple. After making our rocket we made our final motor which we used for the launching of our rocket. We had a sugar motor and we also added iron oxide. We did the same process we did for what we decided our final motor would be (after the tests we had for our motor). We also made a parachute for our rocket (which is in the photos). Looking Beyond: What's the science? While making our rocket, I learned about the center of thrust and mass, and where it should be on a rocket. The center of thrust should be where 50% of the surface are is, which means that the center of thrust should not be in the center of the rocket, but instead slightly above the fins because the fins make up a lot of the surface area. The center of mass should be where 50% of the mass is. This can be measured by finding where the rocket would balance on one finger. The center of thrust should be below the center of mass. If all is good and everything matches up, your rocket should fly great (: Looking Inward: Humans doing science - Lab Practice In the process of making our rocket and final rocket motor, I helped a lot. I would say I was for the most part the leader of the group, and we ended up with a great result. We all worked hard and it showed. Our rocket flew the highest in the whole team, and we were really proud of that. We sadly couldn't find our rocket after and were told it went in the bay. When launching though, a lot of people used injection chargers and my group decided not to.. Since most groups that used it rockets blow up.. So that was a plus for us! Looking Outward: Collaboration and Impact My group throughout this Rocket Project was Rebekah Brandon, and I of course. I've worked with them in many projects, and I always enjoy the collaboration.l Us as a team did great. As I mentioned above, we basically killed it and had the best flying rocket. We all worked hard! Proud of my group! Looking Forward: What next? In the future I would love to try making even bigger rockets and see how far I can take this rocketry. Who knows, maybe I can design rockets that go to the moon, or even further. Take Aways: Rocketry is an actually thing. Below is a slide show showing our process and below that is the video our launch. Sorry in advance, I didn't get the whole thing.
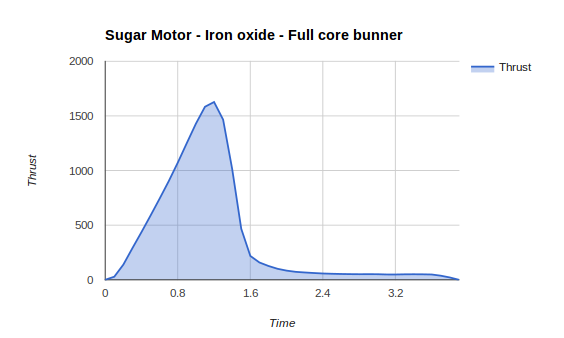
Data Collection Sum of Thrust: 14653.3 Total Impulse: 14.37 newtons Average Thrust: 3.78 newtons After finding our rocket thrust calculations my group and I did calculations on our final rocket. If our rocket weighed 0.1kg and our final motors average thrust was 3.78 newtons and our burn time was 3.8secs, that would mean our final rocket would have a peak altitude of 94.70 meters and 310.70 in feet. If our rockets body radius was .04 meters than our rocket body cross sectional area would be .055 meters.
What we wanted to achieve: When given the assignment of making our own VR Goggles, I was super excited. I chose to work with Rebekah for this assignment and our main goal was to finish our VR goggles ( obviously ) and to create the best 360 degree VR scene we could make. Our design: We chose to go with a simple design we got offline. Below is the design. Construction progress: Making the VR Goggles wasn't a hassle. It was just a simple process of cutting and tapping parts together. We made our VR Goggles out of cardboard and cut that cardboard into pieces from the online design we got. We taped those pieces together and later on got our lenses for the goggles and taped that too to the VR Goggles. Below is pictures of our process. Finished VR Goggle and VR scenes:
FIRST MOTOR: With the first motor, my group (Brandon, Rebekah, and myself) we made it with our Chem teacher. That was so, so that when we did make it by ourselves, we would know what were were doing. Below is a picture of our first motor: SECOND MOTOR: The second motor we made was with supervision. My group and I decide to team up with another group for this one since we were making the same one. Below is a picture of the second motor: FINAL MOTOR: For our third one, we finally got to make it ourselves. We tested our final one and it didn't explore and that was great! Below is pictures of the process: Looking Back - What happened?: For each rocket we pretty much made them exactly the same. We just kept making them to verify what motor we wanted for our final rocket. The only difference with the motors was with our final motor. Instead of adding 25grams of sugar fuel, we added 30grams. Beside that, for each motor we added iron oxide, 25grams of sugar, and each motor was tapped and were full core motors.
Looking Beyond - What do you think has happened? What’s the science?: Throughout each launch we didn't have any of our motors explode and that was a plus! That meant the mixes we had we just right, and even for our final motor, which we add 10 more grams of the fuel. Looking Inward - Humans doing science Lab Practice: I think we got desirable results because none of of motor exploded as I mentions before. A few of my classmates motors did exploded and it was peculiar that it tend to be the ones with Black powder. My take on that was maybe black powder was so strong and adding 25grams of that, plus the cracks in the motor sell itself can add the the exploding? Looking Outward - Collaboration and Impact: For each motor I worked with people that were great! the people I worked with were on the same page I was on and we worked through making the motors in a positive way where by everyone participated. We all strive on making great motors and we did achive that. Looking Forward - What next?: My group (Rebekah and Brandon) plan on making our final rocket and using the final motor as, well our final motor. LET THE LAUNCHING OF ROCKETS BEGIN! Take Away: Rocketry is great. Looking Back - What happened?: Going into this experiment I didn't know what to expect. When we were told we were going to make rocks I didn't think it was going to come out the way it did. First we poured water into two small test tubes, then put sodium silicate in the tubes. After that we poured calcium chloride into one of them, and cobalt chloride into the other. One of the test tubes formed a purple and blue rock, and the other formed a white rock. The longer we left it there, the bigger the rock got.
Looking Beyond - What do you think has happened? What’s the science?: I learned that silicate in the crust creates rocks by bonding with other metals and drying at the surface. Sodium silicate dissolves and form silicate ions as they would be in the mantel, allowing us to introduce metals and make rocks. For this lab, we combined sodium silicate and calcium chloride to get calcium silicate rock and sodium chloride. Looking Inward - Humans doing science Lab Practice: I think my group did a good job because our rocks did form and grow, which I think was the point. We all tried our best to make the most out of the experiment, and it showed. Looking Outward - Collaboration and Impact: For this lab I worked with Rebekah, Brandon, Joey, and Nick. I think we worked good together. We all did equal amount of work and there wasn't really any problem getting people to work. Looking Forward - What next?: I wonder if there's any other way to make the rocks, or any other colors we can make it. I also wish if we had bigger testing tubs. It would have been awesome to see the rocks up close since we could only see so much when it was in the small testing tube. Take Aways: Don't except the littlest. Looking Back - What happened?: For this experiment we made Phee Bombs (bath bombs). We mixed citric acid, corn starch, epsom salts and baking soda, poured water into that mix (little amounts at a time) and added scents that we wanted the bath bombs to be, mixed it to the point where we got a sand type substances. After that, we put that sand substance into two circle covers that you'd get with your Starbucks drink. We put one on top the other and duck taped the whole thing making sure there was no holes or openings all around it. We made two or those and after wrapping we put them in the freezer over night. When we unwrapped them the next day (24 in freezer) one of the Phee Bombs come out in two halfs and the other one was actually a circle.
Looking Beyond - What do you think has happened? What’s the science?: Before actually making the Phee Bombs we did research, and found out the science behind bath bombs and how it worked like it did. Bath bombs are suppose to fizzle and expand throughout your bathtub making it more enjoyable. The fizzling and expanding of the bath bomb is cause because of the mixture of the sodium bicarbonate and citric acid. These chemicals are solid and together when they are dry powders, but in water they dissolve and the citric acid reacts with the sodium bicarbonate to form sodium citrate and carbon dioxide. Looking Inward - Humans doing science Lab Practice: I think our bath bombs came out good because when we put it in the water it did fizzle and expand and looked like the store bought bath bombs. Looking Outward - Collaboration and Impact: For this lab I worked with Rebekah and our collaboration was great! She's one of my closest friends and I liked that when it was work time, it was work time and we got to business. We both did an equal amount of work, and the experiment/experience was great. Looking Forward - What next?: If we were to do this experiment again we would maybe explore other ingredients that we could make the bath bombs with. Take Aways: When it says add little water at a time ADD LITTLE WATER AT A TIME. Today my partner (Rebekah) and I got our Independent Experiment Approved! We plan on starting out Phee Bombs (bath bombs) tomorrow (:
|
Fatuma Fadhil
Throughout this blog I will be posting all things Chemistry. Every post will be something I did/doing in my Chemistry Class |




















 RSS Feed
RSS Feed
